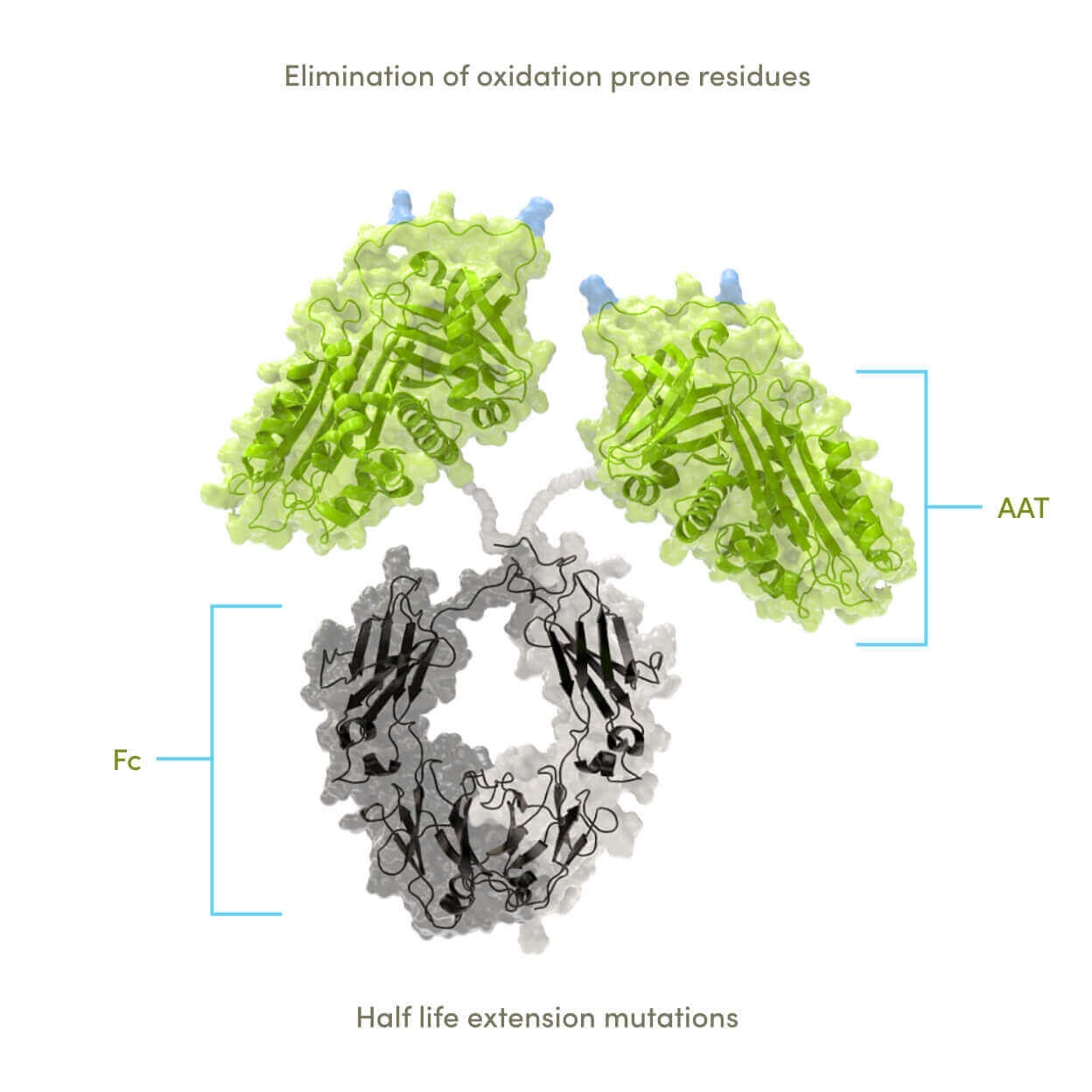
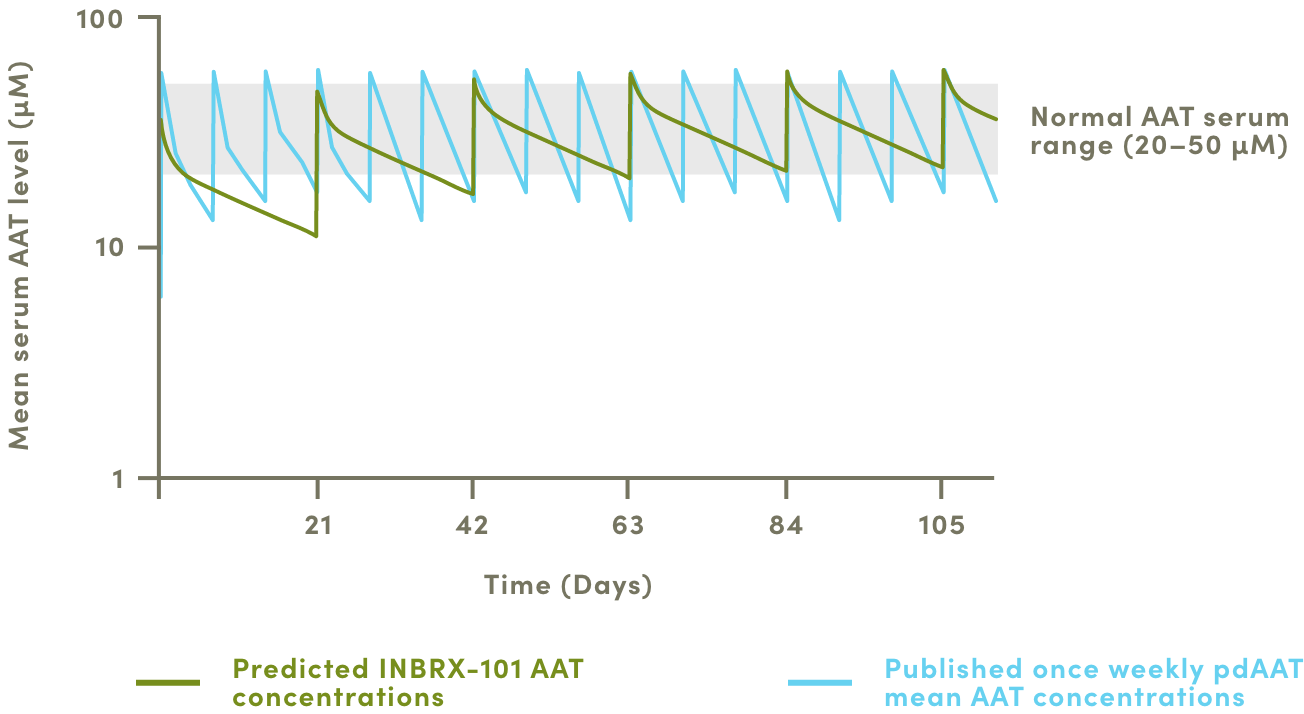
The current state and standard of care for people with AATD is suboptimal. Receiving plasma-derived AAT with weekly infusions doesn’t maintain normal serum AAT levels. AAT has proven difficult to develop recombinantly, often displaying loss of activity and experiencing accelerated degradation.
We believe our novel approach has the potential to overcome these challenges. Fusion of AAT to the Fc region allows for production using a standard antibody expression system, which, when combined with our proprietary AAT-function preserving purification process, generates substantial and potentially scalable yields of active recombinant AAT protein.
We believe that this extended exposure supports the potential for dosing every three or four weeks while maintaining patients in the normal range of AAT exposure, which in turn would reduce the frequency of annual infusions, eliminate lung decline from Alpha-1 Disease, and could significantly improve patient quality of life.